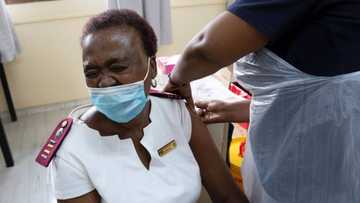
No Fault Compensation Scheme for Covid19 Vaccine Injuries Is in the Works
- Cooperative Governance and Traditional Affairs Minister Nkosazana Dlamini-Zuma has asked the public for their comments on the new vaccine scheme
- The no-fault compensation scheme proposes compensation to South Africans who have suffered an injury due to the administration of the Covid-19 vaccine
- Health Minister Dr Zweli Mkhize revealed that around R250 million has been set aside for the scheme's funding
PAY ATTENTION: Click “See First” under the “Following” tab to see Briefly.co.za News on your News Feed!
COGTA Minister Nkosazana Dlamini-Zuma has asked South Africans for their comments on the no-fault compensation scheme for the Covid-19 vaccine. The scheme proposes providing compensation to people who have suffered a Covid-19 vaccine-related injury.
Dlamini-Zuma stated that the scheme is a vital part of the vaccine rollout programme in the country. The minister revealed that Minister of Health Dr Zweli Mkhize and Minister of Finance Tito Mboweni would be setting an account up for the scheme.
Reports say that the vaccine no-fault compensation scheme will have around R250 million dedicated towards it.
Source: Getty Images
News24 reported that Mkhize stated that the R250 million figure was based on around 800 to 2 000 successful case claims. A report by BusinessTech stated that the money will only cover a person who suffered an injury from a vaccine that is:
- Registered or approved by Sahpra
- Distributed and procured by the South African government
- If one is inoculated in a facility within South Africa
In other news about the Covid-19 vaccine, Briefly News earlier reported that the South African Health Products Regulatory Authority has proposed that the temporary ban on the vaccine be lifted, provided that certain conditions are met. One of these conditions insists on improved screening and monitoring of recipients who pose a high risk of blood clotting.
Sahpra stated on Wednesday that there were no significant safety concerns discovered based on recently reviewed data from Johnson & Johnson’s local research study immunising healthcare workers.
Following reports made by IOL, a US panel will gather again next week to discuss whether the halt on the use of the vaccine should remain. This after six people out of the almost 7 million people who were vaccinated were discovered to have rare cases of blood clots.
Enjoyed reading our story? Download BRIEFLY's news app on Google Play now and stay up-to-date with major South African news!
Source: Briefly News